|
Tunneled Frit Nano-UPLC-MS Column
Omics, Proteomics, Glycomics, Lipidomics,
Metabolomics Mass spectrometer,
LC-MS, LC/MS, nano-HPLCMS, capillary LC column, Trapping colum,...
Mass spectrometer is the key tools for most
Omics research, Such as for
the Proteomics,
Glycomics,
Lipidomics,
Metabolomics, etc.
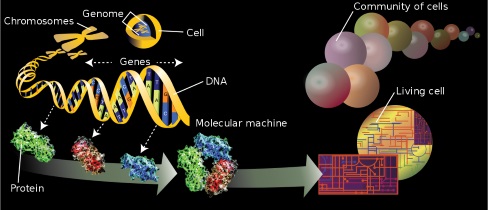
Protein mass spectrometry refers
to the application of mass spectrometry to
the study of proteins.
Mass spectrometry is an important method for the accurate mass determination and
characterization of proteins, and a variety of methods and instrumentations have
been developed for its many uses. Its applications include the identification of
proteins and their post-translational modifications,
the elucidation of protein complexes, their subunits and functional
interactions, as well as the global measurement of proteins in proteomics.
It can also be used to localize proteins to the various organelles, and
determine the interactions between different proteins as well as with membrane
lipids.[1][2].
|

|
|
In combination with protein fractionation, isotopic
labeling and LC-MS, large amounts of protein can be identified and quantified in
a short period of time.
|
Protein and peptide fractionation
Proteins of interest are usually part of a complex mixture of multiple proteins
and molecules, which co-exist in the biological medium. This presents two
significant problems. First, the two ionization techniques used for large
molecules only work well when the mixture contains roughly equal amounts of
constituents, while in biological samples, different proteins tend to be present
in widely differing amounts. If such a mixture is ionized using electrospray or MALDI,
the more abundant species have a tendency to "drown" or suppress signals from
less abundant ones. Second, mass spectrum from a complex mixture is very
difficult to interpret due to the overwhelming number of mixture components.
This is exacerbated by the fact that enzymatic digestion of a protein gives rise
to a large number of peptide products.
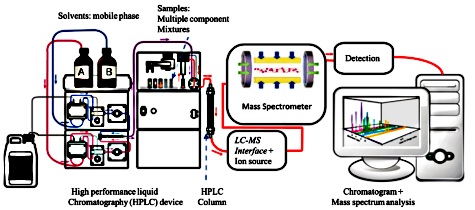
High performance liquid
chromatography is
used to fractionate peptides after enzymatic digestion. Characterization of
protein mixtures using HPLC/MS is also called shotgun proteomics and MuDPIT
(Multi-Dimensional Protein Identification Technology). A peptide mixture that
results from digestion of a protein mixture is fractionated by one or two steps
of liquid chromatography. The eluent from the chromatography stage can be either
directly introduced to the mass spectrometer through electrospray ionization, or
laid down on a series of small spots for later mass analysis using MALDI.
|
|
|
|
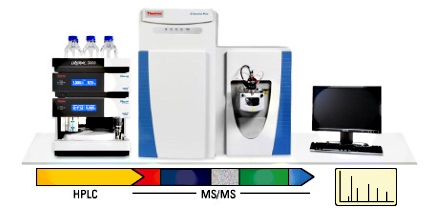
Nano-UHPLC-MS system & Capillary LC column
The complexity and broad dynamic range of the proteome presents great analytical
challenges in global proteomic profiling. Because of its high-sensitivity, high
resolving power, and the robustness of the instrumentation for polypeptide
analysis, the nano-UHPLC-MS system has developed into the major analytical
platform for proteomics research. And among these, the performance of the Fused
Silica Capillary LC column, an essential component in the nano-UHPLC-MS system,
is one of the key to provide high resolution separation of peptides with high
sensitivity.
|

|
|
|
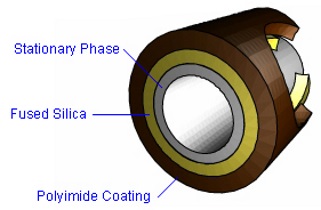
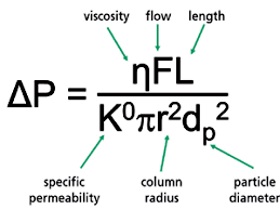
Performance requirements - Capillary LC columns
Trap columns or analytical columns are employed in liquid chromatography
including ultra high pressure systems (UPLC) and in capillary electrophoresis
(CEC) systems to separate and resolve a wide variety of proteins. The 3um or 1.7
um 100 A particles gel packed in the capillary columns of nano-LC-MS or
nano-UUPLC-MS systems, normally result extremely high back-pressure from 5000 to
9000 psi range. The Capillary column for such high pressure systems needs
certain manufacturing characteristics :
|
1. The polyimide coating on the column end has to be maintained to prevent the column from
breaking. under the normally ultra high pressure operations.
|
2. The frit needs high mechanical strength for ultra high pressure resistance.
|
3. Low back pressure of the frit preventing over pressure of the pumps during analysis.
|
|

|
|
Traditional drawback:
|
1. Tapered tip losses the stability and sensitive ionization efficiency, once
broken or contaminated, and need a new one be replaced.
|
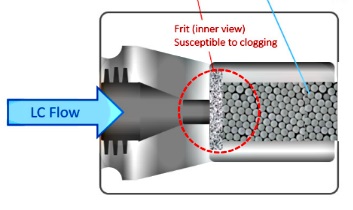
2. Tapered tip cannot be applied to the fabrication of trapping column. |
3. Trap column can often be blocked by un-removed gel species or particles from
Sample solutions after batch analyses or it can lose the concentration factor
after numerous sample analyses. |

|

|
|
|
4. Frequent trap column consumption is an expensive cost item in a proteomics lab that
routinely runs protein samples. |
5. Capillaries becomes fragile at the position of the frit by the removing of
polyimide coating during frit fabrication .
|
6. The frequent clogging, breaking and contamination of the tapered tips or tapered tips with
sintered fits causing the replacement of the entire packed column, however will
limit the use of the packed column with tapered tip. |

|

|
|
|
7. The frit that is formed by dipping the sol-gel Solution to a capillary column with dry Si
resins inside, can not realized good adhesion & homogeneity, and often be
flushed out during the application of high pressure. |
8. Frit strength increased by the capillary action time of the Sol-gel solution in the packed
end can results in extremely high back pressure that is not suitable for high
pressure LC applications. |
|

|
|
|
Advantages of our key patented Tunneled Frit
Nano-UPLC-MS Column
|

|
|
|
1. Easy fabrication process and reproducible.
|
2. Extensive durability. |
3. Low back pressure, High liquid flow, which is suitable for the nanoflow UPLC application. |
|

|
|
|
4. Without any particle leakage or pressure instability demonstrated to sustain 10,000 psi
continuous liquid flow with a short packing (2 cm C18 particle) tunneled-frit
column.
|
5. Successfully applied to the fabrication of nanoUPLC trapping and analytical
column system.
|
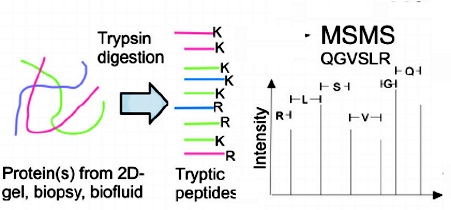
6. Analysis tryptic peptides with high separation efficiency and sensitivity.
|
|

|
|
|
7. Improved detection sensitivity for phosphopeptide analysis that may be interfered by metal
parts.
|
8. Packed column with the frit at one end is durable to run in extreme repeats and long time,
and may be re-packed with new packing materials. |
|
Tunnel Frit: A Nonmetallic
In-Capillary Frit for Nanoflow Ultra High-Performance Liquid ChromatographyMass
Spectrometry Applications
(by courtesy of author & patent inventor)
ABSTRACT:
In this study, an easy method to fabricate a durable in-capillary frit was
developed for use in nanoflow liquid chromatography (nanoLC). A small orifice
was tunneled into the solgel frit during the polymerization process resulting in
the simple fabrication of a tunnel frit. A short packing tunnel frit column (2
cm, C18 particles) was able to sustain over 10 000 psi continuous liquid flow
for 10 days without observation of particle loss, and back pressure variation
was less than 5%. The tunnel frit was successfully applied to the fabrication of
nanoflow ultra high-performance liquid chromatography (nano-UHPLC) trap and
analytical columns. In the analysis of tryptic peptides, the tunnel frit trap
and analytical columns were demonstrated to have high separation efficiency and
sensitivity. In analysis of phosphopeptides, the use of the nonmetallic tunnel
frit column showed better sensitivity than the metallic frit column. This design
can facilitate the preparation of nano-HPLC and nano-UHPLC columns and the
packing material can easily be refilled when the column is severely contaminated
or clogged.
|

|
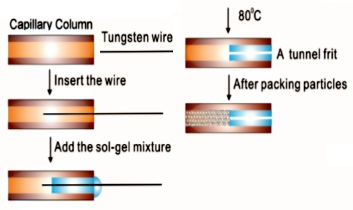
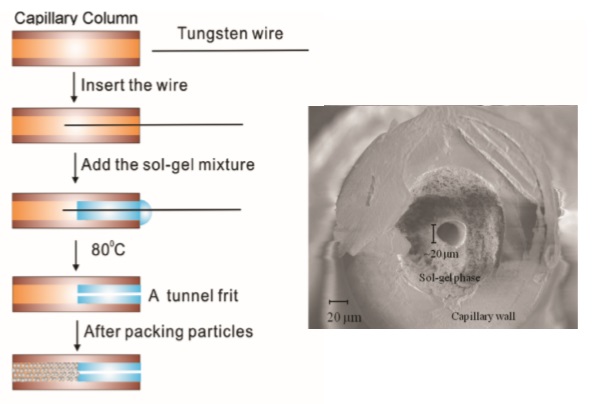
(a) Workflow for the preparation of a tunnel frit capillary.
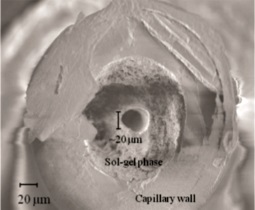
(b) Scanning electron microscopy scan (211�magnification) of an in capillary
tunnel frit made from a 375μm o.d.180μmi.d. capillary and 18 μm tungsten wire to
produce a tunnel. |
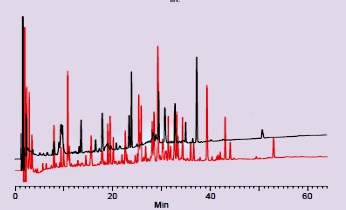
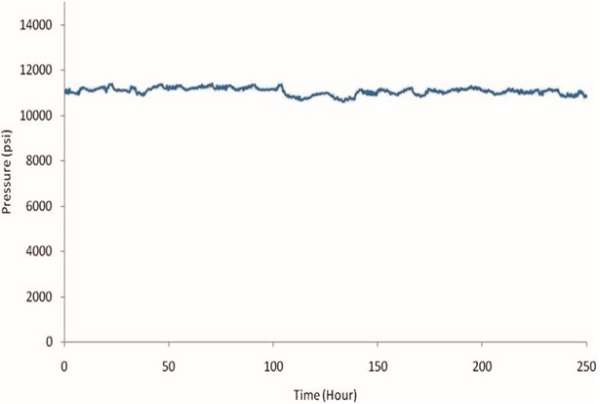
Long-term stability test of the tunnel frit trap column under high pressure
operation with a 170 μL/min flow of H2O/ACN = 99/1 solution.
|
|
|
|
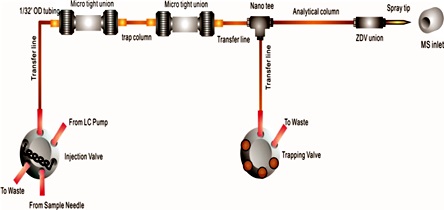
Schematic illustration of the application of the tunnel frit column in a
nano-UHPLCMS system. |
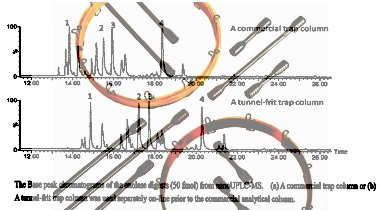
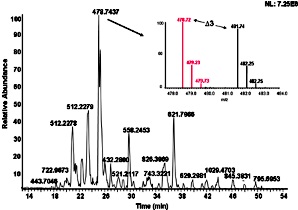
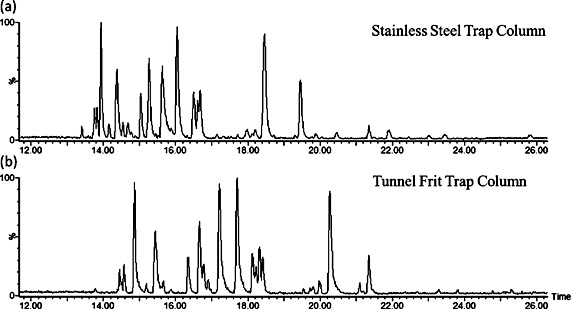
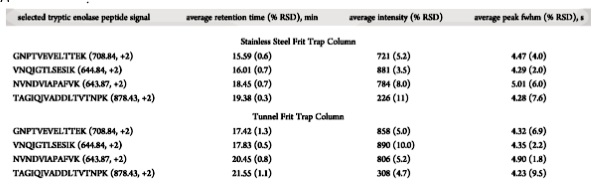
Base peak chromatogram for the analysis of tryptic enolase peptides (50 fmol)
using nano-UHPLCMS with (a) a stainless steel frit or (b) a tunnel frit trap
column coupled with a commercial analytical column. |
|
|
|
Average Retention Time, Intensity and Peak Width of Selected Peptide Signals
from Nano-UHPLCMS Analyses of Tryptic Enolase Peptides (50 fmol) Replicated Six
Times Using a Stainless Steel Frit or Tunnel Frit Trap Column Coupled with a
Commercial Analytical Column |
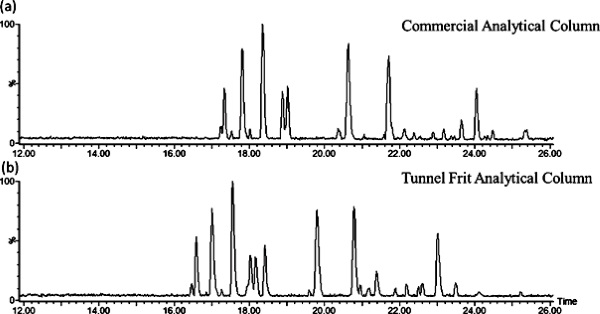
Base peak chromatogram for the analysis of tryptic enolase peptides (50 fmol)
using nano-UHPLCMS with a stainless steel frit trap column coupled with a (a)
commercial or (b) tunnel frit analytical column |
|
|
|
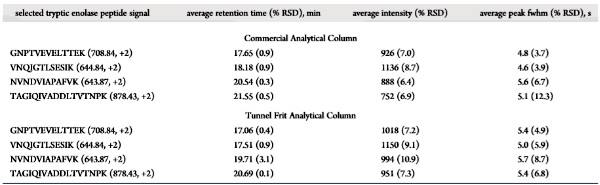
Average Retention Time, Intensity and Peak Width of the Selected Peptide Signals
from Nano-UHPLCMS Analyses of the Tryptic Enolase Peptides (50 fmol) Replicated
Six Times Using a Stainless Steel Trap Column Coupling with a Commercial or
Tunnel Frit Analytical Column |
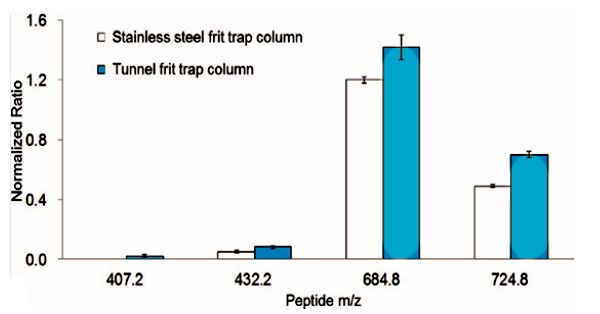
Normalized signal response for four synthetic phosphopeptides (NVPLYpK, m/z =
407.2, 2+; HLADLSpK, m/z = 432.2, 2+; VNQIGTpLSESIK, m/z = 684.8, 2+; and
VNQIGTLSpESpIK, m/z = 724.8, 2+) analyzed using nano-UHPLC�MS with a stainless
steel or tunnel frit trap column coupled with a tunnel frit analytical column.
The nonphosphopeptide peptide signal (NVNDVIAPAFVK, m/z = 643.9, 2+) was used as
an internal standard to normalize the phosphopeptide signal. |

|
|
|
Nano-UHPLC�MS Analysis of TiO2 Purified Phosphopeptides from Jurkat Cell Lysate
Using a Stainless Steel Frit or Tunnel Frit Trap Column Coupled with a Tunnel
Frit Analytical Column |
|
CONCLUSIONS :
In the analysis of protein digestion, the tunnel frit trap
column and the tunnel frit analytical columns produced similar performance to
the commercial system. To our knowledge, this is the first study that has
described an in-capillary frit preparation method for nano-UHPLC applications.
The successful application of our tunnel frit in nano-UHPLCMS
could substantially reduce the cost of the use of trap and analytical columns in
proteomics laboratories. More importantly, this design can improve the
sensitivity of analysis of phosphopeptides. The tunnel frit technique can also
be widely applied to other packed columns for chromatography enrichment or
separation applications.
The TEAM Omics Spectral Library Cooperation Plateform
(TEAM_OSLCP)
The purpose TEAM_OSLCP is to provide an co-operational Omics
Spectral Library build-up entity, composed of its global members applying our
Tunneled Frit Nano-UPLC-MS Column for Omics research and clinical applications,
as for Proteomics, Glycomics, Lipidomics, Metabolomics etc.
The TEAM_OSLCP invite all our Tunneled Frit Nano-UPLC-MS
Column customer to participate the plate form for persistent uploading their
spectral raw data and annotation information into the library.
TEAM_OSLCP provides very helpful Feed Back mechanism for the
members who have significantly participated and uploaded their Omics spectrum to
make the Omics Spectral Library more abundant, comprehensive, and also more
specific.
The Feed Back may includes bonus that mainly are proportion
to the amount of the uploaded entries and the counts they are referenced by
others.
TEAM_OSLCP also initiate for the collection and integration
of utility software, tool software modules and application software, mobile app
that are developed by our members.
Base on the high performance and patented Tunneled Frit
Nano-UPLC-MS Column, our members will not only have very helpful application for
their Omics study, but will also they have the global extendable development
opportunities and resources form their participations and co-operation from our
TEAM_OSLCP.
|
|
|
|
|
|
|