|
Vaccine / Animal
Vaccine Overviews
https://en.wikipedia.org/wiki/Vaccine

|
A vaccine is a biological preparation that provides active acquired
immunity to a particular disease. vaccine typically contains an agent that
resembles a disease-causing microorganism and is often made from weakened or
killed forms of the microbe, its toxins or one of its surface proteins. agent stimulates the body's immune
system to recognize the agent as a threat,
destroy it, and keep a record of it so that the immune system can more easily
recognize and destroy any of these microorganisms that it later encounters.
can be prophylactic (example: to prevent or ameliorate the effects of a
future infection by any natural or "wild" pathogen), or therapeutic (e.g., vaccines
against cancer are being investigated). |
The administration of vaccines is called vaccination.
The effectiveness of vaccination has been widely studied and verified; for
example, the influenza
vaccine,the HPV
vaccine, and the chicken pox vaccine.
Vaccination is the most effective method of preventing infectious diseases; widespread
immunity due to vaccination is largely responsible for the worldwide
eradication of smallpox and the restriction of diseases such
as polio, measles, and tetanus from much of the world. The World Health
Organization (WHO) reports that
licensed vaccines are currently available to prevent or contribute to the
prevention and control of twenty-five infections
The terms vaccine and vaccination are derived from Variolae vaccinae (smallpox of the cow), the term
devised by Edward Jenner to denote cowpox. He used it in 1798 in
the long title of his Inquiry into
the...Variolae vaccinae...known...[as]...the Cow Pox, in which he described
the protective effect of cowpox against smallpox. In 1881, to
honor Jenner, Louis Pasteur proposed that the terms should be
extended to cover the new protective inoculations then being developed.
The Strange History of Vaccines
(The
Strange History of Vaccines—And Why People Fear Them)
|
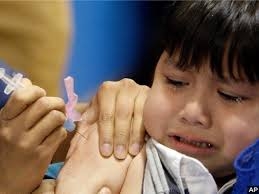
Before vaccines, millions of children died horrific
deaths each year from infectious diseases like whooping cough, polio, and
measles. Today, thanks to vaccines, most of these diseases have been eradicated.
Yet people in different corners of the world are rejecting vaccines. In the
United States, more and more parents are refusing to have their children
vaccinated because they believe a debunked theory that vaccines cause autism.
Meanwhile, in Pakistan and Afghanistan, health workers are regularly targeted
because vaccines are thought to be a Western plot ..... |
|
List of epidemics
https://en.wikipedia.org/wiki/List_of_epidemics
|
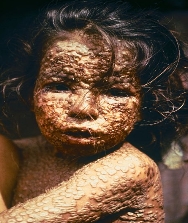
Smallpox is
believed to have been acquired by humans originally as a zoonosis from a terrestrial African rodent between 16,000 and
68,000 years ago, well before the dawn of agriculture and civilization. The
earliest physical evidence of it is probably the pustular rash on the mummified
body of Pharaoh Ramses V of Egypt disease killed an
estimated 400,000 Europeans annually during the closing years of the 18th
century (including five reigning monarchs), and was
responsible for a third of all blindness all those infected, 20–60 percent—and over 80 percent of infected
children—died from the disease. |
|

|
Smallpox
https://en.wikipedia.org/wiki/Smallpox
Smallpox was responsible for an estimated 300–500 million
deaths during the 20th century. As recently
as 1967, the World Health
Organization (WHO) estimated that
15 million people contracted the disease and that two million died in that year.
After vaccination campaigns throughout the 19th and 20th
centuries, the WHO certified the global
eradication of smallpox in 1979. Smallpox is
one of two infectious diseases to have been eradicated, the other
being rinderpest, which was
declared eradicated in 2011.
Veterinary medicine
https://en.wikipedia.org/wiki/Vaccine#Veterinary_medicine

|
Vaccinations of animals are used both to prevent their
contracting diseases and to prevent transmission of disease to humans.[82] Both animals
kept as pets and animals raised as livestock are routinely vaccinated. In some
instances, wild populations may be vaccinated. This is sometimes accomplished
with vaccine-laced food spread in a disease-prone area and has been used to
attempt to control rabies in raccoons.
Where rabies occurs, rabies vaccination of dogs may be required by law. Other
canine vaccines include canine distemper, canine parvovirus, infectious
canine hepatitis, adenovirus-2, leptospirosis, bordatella, canine parainfluenza virus, and Lyme disease, among
others.
|
Cases of veterinary vaccines used in humans have been
documented, whether intentional or accidental, with some cases of resultant
illness, most notably with brucellosis. However, the
reporting of such cases is rare and very little has been studied about the
safety and results of such practices. With the advent of aerosol vaccination in
veterinary clinics for companion animals, human exposure to pathogens that are
not naturally carried in humans, such as Bordetella
bronchiseptica, has likely increased in recent years.In some
cases, most notably rabies, the parallel
veterinary vaccine against a pathogen may be as much as orders of magnitude more economical than the human one.
Animal Vaccine Overviews

Avian reoviruses
禽呼腸孤病毒
Newcastle disease
新城疫病
Infectious bursal disease 傳染性囊腫疾病
Influenza A virus H5N6 subtype
A型流感病毒的H5N6亞型
Japanese encephalitis
日本腦炎
Classical swine fever
典型豬瘟
Bovine ephemeral fever 牛暫時熱
Bovine herpesvirus 1
牛皰疹病毒1
Infectious bovine rhinotracheitis
傳染性牛鼻氣管炎 |
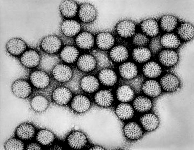
Avian reoviruses
禽呼腸孤病毒
https://en.wikipedia.org/wiki/Avian_reovirus
|
Avian reoviruses belong
to the genus Orthoreovirus,
and Reoviridae family.
They are non-enveloped viruses that undergo replication in the cytoplasm of
infected cells. It has icosahedral symmetry and contains a double-shelled
arrangement of surface protein. Virus particles can range between 70–80 nm.
Morphologically, the virus is a double stranded RNA virus that is composed of
ten segments. The genome and proteins that are encoded by the genome can be
separated into three different sizes ranging from small, medium, or large. Of
the eleven proteins that are encoded for by the genome, two are nonstructural,
while the remaining nine are structural. |
Newcastle disease 新城疫病
https://en.wikipedia.org/wiki/Newcastle_disease
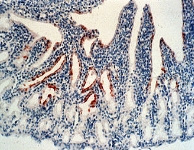
|
Newcastle disease is a contagious bird disease affecting many domestic and wild avian
species; it is transmissible
to humans.It was first
identified in Java, Indonesia, in 1926, and in 1927, in Newcastle-upon-Tyne,
England (whence it got its name). However, it may have been prevalent as early
as 1898, when a disease wiped out all the domestic fowl in northwest Scotland.[2] Its effects
are most notable in domestic poultry due to their high susceptibility and
the potential for severe impacts of an epizootic on the poultry industries. It is endemic to many countries.
Exposure of humans to infected birds (for example in poultry processing plants)
can cause mild conjunctivitis and influenza-like symptoms, but
the Newcastle disease virus (NDV) otherwise poses no hazard to human health.
Interest in the use of NDV as an anticancer agent has arisen from the ability of
NDV to selectively kill human tumour cells with limited toxicity to normal
cells.
No treatment for NDV exists, but the use of prophylactic
vaccines and sanitary
measuresreduces the likelihood of outbreaks.
|
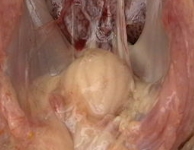
Infectious bursal disease 傳染性囊腫疾病
https://en.wikipedia.org/wiki/Infectious_bursal_disease
|
Infectious bursal disease (also
known as IBD, Gumboro Disease, Infectious Bursitis and Infectious Avian
Nephrosis) is a highly contagious disease of young chickens caused
by infectious
bursal disease virus (IBDV),[1]characterized
by immunosuppression and
mortality generally at 3 to 6 weeks of age. The disease was first discovered in Gumboro, Delaware in
1962. It is economically important to the poultry industry worldwide due to
increased susceptibility to other diseases and negative interference with
effective vaccination.
In recent years, very virulent strains of IBDV (vvIBDV), causing severe
mortality in chicken, have emerged in Europe, Latin America, South-East Asia, Africa and
the Middle East.
Infection is via the oro-fecal route, with affected bird excreting high levels
of the virus for approximately 2 weeks after infection. |
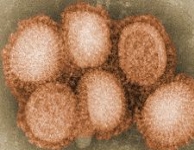
Influenza A virus H5N6 subtype A型流感病毒的H5N6亞型
https://en.wikipedia.org/wiki/Influenza_A_virus_subtype_H5N6
|
H5N6 is a subtype of the species Influenza
A virus (sometimes called bird
flu virus). Four known cases, three fatal,
have occurred in humans as of July 12, 2015.2016
In 2016 cases of H5N6 were reported alongside H5N8, and H7N9 across the globe.Today, 22 of November 2016, South Korea called for many H5N6. Many cases were
reported.
In November and December human cases of H5N6 were reported in China. In bird, by December there were four outbreaks in China
since October and forced the culling of more than 170,000 birds.2016 South Korea had raised its
bird flu alert to highest level for the first time.The heightened alarm status came as the country grappled
with an outbreak of the highly pathogenic H5N6 bird flu that started a month ago
in November. By the start of December H5N6 avian influenza was reported in bird
droppings in Hong Kong. |
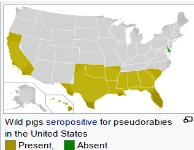
Aujeszky's disease(seudorabies)
Aujeszky病(偽狂犬病)
https://en.wikipedia.org/wiki/Pseudorabies
|
Aujeszky's disease, usually
called pseudorabies in the United States, is a viral disease in swine that has been endemic in most parts of the world. It is
caused by Suid herpesvirus 1 (SuHV1). Aujeszky's disease is
considered to be the most economically important viral disease of swine in areas
where hog cholera has been eradicated. Other mammals, such as humans, cattle, sheep, goats, cats, dogs, and raccoons, are also
susceptible. The disease is usually fatal in these animal species bar humans.
The term "pseudorabies" is found inappropriate by many people, as SuHV1 is a herpesvirus and not related to the rabies virus.
Research on SuHV1 in pigs has pioneered animal disease
control with genetically modified vaccines. SuHV1 is now used in model studies
of basic processes during lytic herpesvirus infection, and for unravelling
molecular mechanisms of herpesvirus neurotropism.
|
Japanese encephalitis 日本腦炎
https://en.wikipedia.org/wiki/Japanese_encephalitis
Classical swine
fever 典型豬瘟

|
Classical swine fever (CSF)
or hog
cholera (also
sometimes called pig
plague based
on the German word Schweinepest)
is a highly contagious disease of swine (Old World and New World pigs).
Virus
The infectious agent responsible is a virus CSFV (previously called hog cholera
virus) of the genus Pestivirus in the family Flaviviridae. CSFV is
closely related to the ruminant pestiviruses that cause bovine viral diarrhoea
and border disease.[6]
The effect of different CSFV strains varies widely, leading to a wide range of
clinical signs. Highly virulent strains correlate with acute, obvious disease
and high mortality, including neurological signs and hemorrhages within the skin.
Less virulent strains can give rise to subacute or chronic infections that may
escape detection, while still causing abortions and stillbirths. In these cases,
herds in high-risk areas are usually serologically tested on a thorough
statistical basis.
Infected piglets born to infected but subclinical sows help maintain the disease
within a population. Other signs can include lethargy, fever, immunosuppression,
chronic diarrhoea, and secondary respiratory infections. The incubation period
of CSF ranges from 2 to 14 days, but clinical signs may not be apparent until
after 2 to 3 weeks. Preventive state regulations usually assume 21 days
|
Bovine ephemeral fever 牛暫時熱
https://en.wikipedia.org/wiki/Bovine_ephemeral_fever

|
Bovine ephemeral fever (BEF)
also known as Three Day Sickness is an arthropod vector-borne
disease of cattle and is caused by bovine ephemeral fever virus (BEFV), a member
of the genus Ephemerovirus in
the family Rhabdoviridae.
Virology
BEFV forms a bullet- or cone-shaped virions that consist of a negative, single
stranded RNA genome with a lipid envelope and 5 structural proteins. The
envelope glycoprotein G contains type-specific and
neutralizing antigenic sites. There has been recent evidence which demonstrated
that BEFV induces apoptosis in several cell lines. It was however
shown that apoptosis could be blocked by the caspase inhibitor (Z-VAD-fmk), indicating that
BEFV induces caspase-dependent apoptosis in cultured cells.
|
Bovine herpesvirus 1 牛皰疹病毒1
https://en.wikipedia.org/wiki/Bovine_herpesvirus_1
|
Bovine herpesvirus 1 (BoHV-1)
is a virus of the family Herpesviridae and
the subfamily Alphaherpesvirinae,
known to cause several diseases worldwide in cattle,
including rhinotracheitis, vaginitis, balanoposthitis, abortion, conjunctivitis,
and enteritis.
BoHV-1 is also a contributing factor in shipping fever,
also known as bovine
respiratory disease (BRD).
It is spread horizontally through
sexual contact, artificial
insemination,
and aerosol transmission and it may also be transmitted vertically across
the placenta. BoHV-1 can cause both clinical and subclinical infections,
depending on the virulence of
the strain. Although these symptoms are mainly non-life-threatening it is an
economically important disease as infection may cause a drop in production and
affect trade restrictions. Like other herpesviruses, BoHV-1 causes a lifelong
latent infection and sporadic shedding of the virus. The sciatic nerve and trigeminal nerve are
the sites of latency.
A reactivated latent carrier is normally the source of infection in a herd. The
clinical signs displayed are dependent on the virulence of the strain. There is
a vaccine available
which reduces the severity and incidence of disease. Some countries in Europe
have successfully eradicated the
disease by applying a strict culling policy.
|
Infectious bovine rhinotracheitis 傳染性牛鼻氣管炎
|
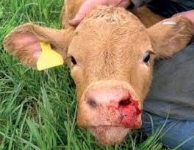
Infectious bovine rhinotracheitis
The respiratory disease caused by BoHV-1 is commonly
known as infectious bovine rhinotracheitis. This disease affects the upper
respiratory tract as well as the reproductive tract of cattle, and is commonly
found in feedlots across North America.
Clinical symptoms include fever, serous to mucopurulent
nasal discharge, coughing, sneezing, difficulty breathing, conjunctivitis and loss of appetite. Ulcers commonly
occur in the mouth and nose. Mortality may reach 10 percent.
IBR can also cause abortion. This generally
occurs in mid-gestation when a susceptible cow is infected with BoHV-1. A viraemia occurs and subsequently the virus
crossed the placenta and causes organ necrosis in the fetus. BoHV-1 also causes a
generalized disease in newborn calves, characterized by enteritis and death.
|
|
|
Porcine epidemic diarrhea virus
豬流行性腹瀉病毒
Porcine reproductive and respiratory syndrome virus
豬繁殖與呼吸綜合徵病毒Porcine
circovirus
豬圓環病毒
Classical swine fever
典型豬瘟
Streptococcus suis
鏈球菌
Actinobacillus pleuropneumoniae
放線桿菌胸膜肺炎Erysipelothrix
rhusiopathiae
豬丹毒絲狀菌
Pasteurella multocida
多殺性巴斯德桿菌Pasteurella
巴氏桿菌
Coccidiosis
球蟲病
Listeria
李斯特菌
Listeriosis
李斯特菌病
Derzsy病
|
Porcine epidemic
diarrhea virus 豬流行性腹瀉病毒

|
Porcine epidemic diarrhea virus (PED virus or PEDV) is a coronavirus that infects the cells lining the
small intestine of a pig, causing porcine
epidemic diarrhoea, a condition of severe diarrhea and dehydration. Older hogs
mostly get sick and lose weight after being infected, where as newborn piglets
usually die within five days of contracting the virus. PEDV cannot be
transmitted to humans, nor contaminate the human food supply.
It was first discovered in Europe, but has become
increasingly problematic in Asian countries, such as Korea, China, Japan, the Philippines, and Thailand. It has also spread
to North America: it was discovered in the United States on May 5, 2013 in Indiana, and in Canada in the winter of 2014. In January
2014, a new variant strain of PEDV with three deletions, one insertion, and
several mutations in S (spike) 1 region was identified in Ohio by the Animal
Disease Diagnostic Lab of Ohio Department of Agriculture.
PEDV has a substantial economic burden given that it is
highly infectious, resulting in significant morbidity and mortality in piglets. Morbidity and mortality
rates were lower for vaccinated herds than for nonvaccinated herds, which
suggests the emergence of a new PEDV field strain(s) for which the current
vaccine, based on the CV777 strain, was partially protective. Consumers are
likely to feel the effects of the viral disease in the form of higher prices for
pork products.
|
Porcine reproductive and respiratory syndrome
virus
豬繁殖與呼吸綜合徵病毒
https://en.wikipedia.org/wiki/Porcine_reproductive_and_respiratory_syndrome_virus
|
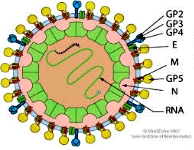
Porcine reproductive and respiratory syndrome virus (PRRSV)
is a virus that
causes a disease of pigs,
called porcine
reproductive and respiratory syndrome (PRRS),
also known as blue-ear
pig disease (in Chinese, zhū
láněr bìng 豬藍耳病).
This economically important, panzootic disease
causes reproductive failure in breeding stock and respiratory tract illness in
young pigs. Initially referred to as "mystery swine disease" and "mystery
reproductive syndrome," it was first reported in 1987 in North America (2)
and Central Europe (3).
The disease costs the United States swine industry around $644 million annually,
and recent estimates in Europe found that it costs almost 1.5b€ every year.
Porcine Reproductive and Respiratory Syndrome (PRRS) is a complex disease.
Modified Live Vaccines (MLV) vaccines are the primary immunological tool for its
control, but PRRS control goes way beyond than just vaccination, and in order to
achieve sustainable results, a systematic approach should be implemented. It
requires a full understanding of the disease and a set of tools to achieve a
long term success, therefore a standardized 5 step process
has
been developed to successfully achieve PRRS control. A strong platform to consolidate PRRS control in pig farms, large production systems and even
geographical areas has been developed. This platform is a pig population
approach having as main goals: - to maximize immunity, - reduce PRRS virus
(PRRSv) exposure and - prevent new PRRSv infections. The Complexity of PRRS has
allowed implementing successfully this methodology in the Swine Industry around
the globe. |
Porcine circovirus 豬圓環病毒
https://en.wikipedia.org/wiki/Porcine_circovirus
|
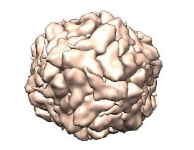
Porcine circovirus(PCV) is a single-stranded DNA virus (class II), that is
nonenveloped with an unsegmented circular genome. The viral
capsid is icosahedral and approximately 17 nm in diameter. PCV is a member of
the virus family Circoviridae.
PCVs are the smallest viruses replicating autonomously in eukaryotic cells.They
replicate in the nucleus of infected cells, using the host polymerase for genome
amplification.
There are 2 strains: type 1 and type 2. Porcine
Circovirus Associated Disease is
caused by porcine circovirus type 2 (PCV2).
PCV-1 (first identified in 1974) readily infects, but is not known to cause
disease in swine; the type 2 has caused problems in recent years with the
increasing occurrence of postweaning
multisystemic wasting syndrome (PMWS),
which over time results in significant depletion of lymphocytes; postmortem
examination of diseased animals reveals enlarged lymph nodes and abnormal lung tissue.
|
Classical swine fever 典型豬瘟
https://en.wikipedia.org/wiki/Classical_swine_fever

|
Classical swine fever(CSF)
or hog
cholera (also
sometimes called pig
plague based
on the German word Schweinepest)
is a highly contagious disease of swine (Old World and New World pigs).
The infectious agent responsible is a virus CSFV (previously called hog cholera
virus) of the genus Pestivirus in the family Flaviviridae.CSFV is
closely related to the ruminant pestiviruses that cause bovine viral diarrhoea
and border disease.
The effect of different CSFV strains varies widely, leading to a wide range of
clinical signs. Highly virulent strains correlate with acute, obvious disease
and high mortality, including neurological signs and hemorrhages within the skin.
Less virulent strains can give rise to subacute or chronic infections that may
escape detection, while still causing abortions and stillbirths. In these cases,
herds in high-risk areas are usually serologically tested on a thorough
statistical basis.
Infected piglets born to infected but subclinical sows help maintain the disease
within a population. Other signs can include lethargy, fever, immunosuppression,
chronic diarrhoea, and secondary respiratory infections. The incubation period
of CSF ranges from 2 to 14 days, but clinical signs may not be apparent until
after 2 to 3 weeks. Preventive state regulations usually assume 21 days
|
Streptococcus suis 鏈球菌
https://en.wikipedia.org/wiki/Streptococcus_suis
|
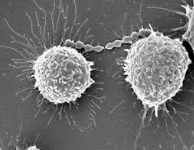
Streptococcus suis is a peanut-shaped, Gram-positive bacterium, and an important pathogen of pigs. Endemic in nearly all countries with an
extensive pig industry, S. suis is also a zoonotic disease, capable of
transmission to humans from pigs.
Humans can be infected with S. suis when they handle infected pig
carcasses or meat, especially with exposed cuts and abrasions on their hands.
Human infection can be severe, with meningitis, septicaemia, endocarditis, and deafness as possible outcomes of infection. Fatal cases
of S. suis are uncommon, but not unknown.
Penicillin is the most common antibiotic used in treatment of S. suis infection; in cases with cardiac
involvement (endocarditis), gentamicin should also be given for synergistic
effect.
|
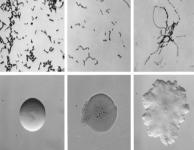
Actinobacillus pleuropneumoniae 放線桿菌胸膜肺炎
|
Actinobacillus pleuropneumoniae (previously Haemophilus
pleuropneumoniae),
is a Gram-negative, facultative anaerobic,
respiratory pathogen found
in pigs.
It was first reported in 1957, and was formally declared to be the causative
agent of porcine pleuropneumonia
in 1964. It
was reclassified in 1983 after DNA studies showed it was more closely related to A.
lignieresii.
A. pleuropneumoniae is
a nonmotile, Gram-negative, encapsulated coccobacillus bacterium found
in the Pasteurellaceae family. It
exhibits β-hemolysis activity, thus
explaining its growth on chocolate or blood agar,
but must be supplemented with NAD ('V factor') to facilitate growth for one of
its biological variants (biovar 1). As
a facultative anaerobic pathogen, A.
pleuropneumoniae may
need CO2 to
grow. Depending
on the biovar, the bacteria may or may not be positive for urease;
both biovars are positive for porphyrin. |
Erysipelothrix rhusiopathiae
豬丹毒絲狀菌
https://en.wikipedia.org/wiki/Erysipelothrix_rhusiopathiae
|
Erysipelothrix rhusiopathiae is a Gram-positive,
catalase-negative, rod-shaped, nonspore-forming, nonacid-fast, nonmotile bacterium. The organism was
first established as a human pathogen late in the 19th century. It may be
isolated from soil, food scraps, and water contaminated by infected animals. It
can survive in soil for several weeks. In pig faeces, the survival period of
this bacterium ranges from 1 to 5 months It grows
aerobically and anaerobically and does not contain endotoxin. Distributed
worldwide, E. rhusiopathiae is primarily considered an animal pathogen, causing a disease
known as erysipelas in animals (and erysipeloid in humans – see below). Turkeys and pigs are most commonly affected, but cases
have been reported in other birds, sheep, fish, and reptiles In pigs, the
disease is known as "diamond skin disease". The human disease called erysipelas is not caused by E. rhusiopathiae, but by
various members of the genus Streptococcus.
It is most frequently associated as an occupational disease of butchers.
|
Pasteurella multocida
多殺性巴斯德桿菌
|
Pasteurella multocida is
a Gram-negative,
nonmotile, penicillin-sensitive coccobacillus belonging
to the Pasteurellaceae family. Strains
belonging to the species are currently classified into five serogroups (A,
B, D, E, F) based on capsular composition and 16 somatic serovars (1-16). P.
multocida is
the cause of a range of diseases in mammals and birds, including fowl cholera in
poultry, atrophic rhinitis in pigs, and bovine hemorrhagic septicemia in
cattle and buffalo. It can also cause a zoonotic infection
in humans, which typically is a result of bites or scratches from domestic pets.
Many mammals (including domestic cats and dogs) and birds harbor it as part of
their normal respiratory microbiota.
See: Pasteurellosis
P. multocida causes a range of diseases in wild and domesticated
animals, as well as humans. The bacterium can be found in birds, cats, dogs, rabbits,
cattle, and pigs. In birds, P.
multocida causes avian or fowl
cholera disease; a significant disease present in commercial and domestic
poultry flocks worldwide, particularly layer flocks and parent breeder flocks. P. multocida strains that cause fowl cholera in
poultry typically belong to the serovars 1, 3, and 4. In the wild, fowl cholera
has been shown to follow bird migration routes, especially of snow geese. The P. multocida serotype-1 is most associated with
avian cholera in North America, but the bacterium does not linger in wetlands
for extended periods of time. P. multocida causes atrophic rhinitis in pigs; it also can
cause pneumonia or bovine
respiratory disease in cattle. In humans, P. multocida is the most common cause of infection
from wound infections after dog or cat bites. The infection usually shows as
soft tissue inflammation within 24 hours. High leukocyte and neutrophil counts are typically observed, leading
to an inflammatory reaction at the infection site (generally a diffuse,
localized cellulitis). It can also
infect other locales, such as the respiratory tract, and is known to cause
regional lymphadenopathy (swelling of the lymph nodes). In more serious cases, a bacteremia can result, causing an osteomyelitis or endocarditis. The
bacteria may also cross the blood–brain barrier and cause meningitis.
|
Pasteurella 巴氏桿菌
|
Pasteurella is a genus of Gram-negative, facultatively
anaerobic bacteria. Pasteurella species are nonmotile and pleomorphic,
and often exhibit bipolar staining ("safety pin" appearance). Most species are catalase- and oxidase-positive.The genus is
named after the French chemist and microbiologist, Louis Pasteur, who first
identified the bacteria now known as Pasteurella
multocida as the agent of chicken cholera. Pathogenesi
Many Pasteurella species are zoonotic pathogens, and humans can acquire an
infection from domestic animal bites. In cattle,
sheep, and birds, Pasteurella species can cause a life-threatening pneumonia; in cats and dogs,
however, Pasteurella is not a cause of disease, and
constitutes part of the normal flora of the nose
and mouth Pasteurella
haemolytica is a species that
infects mainly cattle and horses: P. multocida is the most frequent causative agent
in human Pasteurella infection Common
symptoms of pasteurellosis in humans include swelling, cellulitis, and bloody
drainage at the site of the wound. Infection may progress to nearby joints,
where it can cause further swelling, arthritis, and abscesses
Pasteurella spp. are generally susceptible to chloramphenicol, the penicillins, tetracycline, and the macrolides
The common occurrence of the bacteria is a reason to be
medically proactive and defensive (antibacterial treatments are often necessary)
if a bite occurs.
|
coccidiosis 球蟲病

|
Coccidiosis is a parasitic disease of the intestinal tract of animals
caused by coccidian protozoa. The disease spreads
from one animal to another by contact with infected feces or ingestion of infected tissue. Diarrhea, which may become
bloody in severe cases, is the primary symptom. Most animals infected with
coccidia are asymptomatic, but young
or immunocompromised animals may suffer severe symptoms and
death.
While coccidia can infect a wide variety of animals,
including humans, birds, and livestock, they are usually
species-specific. One well-known exception is toxoplasmosis caused by Toxoplasma
gondii.
Humans may first encounter coccidia when they acquire a young puppy or kitten
that is infected. Other than T.
gondii, the infectious organisms are canine and feline-specific and are not contagious to humans,
unlike the zoonotic diseases.
|
Listeria 李斯特菌
https://en.wikipedia.org/wiki/Listeria
|
Listeria is a genus of bacteria that, until 1992, contained 10 known
species, each
containing two subspecies. As of 2014, another five species were identified Named after
the British pioneer of sterile surgery Joseph
Lister, the genus received its current name in 1940. Listeria species are gram-positive, rod-shaped, and facultatively
anaerobic, and do not produce endospores The major human pathogen in the Listeria genus is L. monocytogenes.
It is usually the causative agent of the relatively rare bacterial disease listeriosis, a serious infection caused by eating food contaminated with the bacteria. The disease affects pregnant women, newborns, adults with weakened immune systems,
and the elderly.
Listeriosis is a serious disease for humans; the overt
form of the disease has a case-fatality rate around 20%. The two main clinical
manifestations are sepsis and meningitis. Meningitis is
often complicated by encephalitis, when it is
known as meningoencephalitis,
a pathology that is unusual for bacterial infections. L. ivanovii is a pathogen of mammals, specifically ruminants, and has rarely
caused listeriosis in humans.The incubation
period can vary between 3 and 70 days.
|
Listeriosis 李斯特菌病

|
Listeriosis is an infectious but not contagious disease caused by the bacterium Listeria
monocytogenes, far more common in domestics animals (domestic mammals and poultry), especially ruminants, than in human beings. It can also
occur in feral animals—among others, game animals—as well as
in poultry and other birds.
The causative bacterium lives in the soil and in poorly
made silage and is acquired by ingestion. It is
not contagious; over the course of 30-year observation period of sheep disease
in Morocco, the disease only
appeared in the late 2000s (decade) when feeding bag-ensiled corn became common In Iceland, the disease is called
"silage sickness"
The disease is usually sporadic, but can occur as farm outbreaks in ruminants.
Three main forms are usually recognized throughout the affected species:
Listeriosis in animals can rarely be cured with antibiotics (tetracyclines, chloramphenicol and benzyl penicillin also) when
diagnosed early, in goats, for example, by treating upon first noticing the
disease's characteristic expression in the animal's face, but is
generally fatal.
|
Derzsy病 27s_disease
|
Derzsy's disease is caused by a virus from the Parvoviridae family. It affects geese and Muscovy ducks.
The virus is shed in the faeces and thus transmission is horizontal, via the direct faecal-oral route and also indirectly via fomites. Vertical
transmission is also possible.
Clinical disease only occurs in
young geese and ducks between birth and 4–5 weeks of age.
Epidemiology
Several genotypes have been
described.The genotype
is based upon the sequence of the VP3 protein.
Clinical signs and diagnosis
Acute disease leads to death in most birds between the
ages of 7–10 days. Clinical signs are quite limited in those cases. Older
animals tend to show severe systemic and neurological signs and diarrhoea.
Adults do not show any clinical signs.
Viral isolation should be attempted for diagnosis, and immunofluorescence and electron microscopy can confirm the viral infection.
Pathological changes may also help the diagnosis.
|
|
Toxoplasma gondii
弓形蟲 Vibrio Anguillarum
鰻弧菌
Vibrio harveyi
哈維弧菌 CpG Oligodeoxynucleotides CpG寡脫氧核苷酸
DNA vaccination
DNA疫苗接種 |
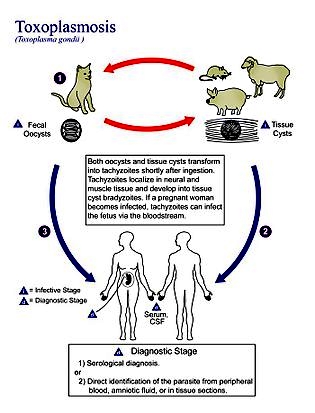
Toxoplasma gondii 弓形蟲
https://en.wikipedia.org/wiki/Toxoplasma_gondii
|
Toxoplasma gondii (IPA ) is an obligate intracellular, parasitic alveolate that causes the disease toxoplasmosis. Found worldwide, T.
gondii is capable of infecting
virtually all warm-blooded animals, but felids such as domestic cats are the only known definitive hosts in which the parasite can undergo
sexual reproduction.
In humans, T.
gondii is one of the most common
parasites in developed countries; serological studies estimate that 30–50% of the global population has
been exposed to and may be chronically infected with T. gondii, although infection
rates differ significantly from country to country. For example, previous estimates have shown the highest
prevalence of persons infected to be in France, at 84%.[10] Although mild, flu-like symptoms occasionally occur during
the first few weeks following exposure, infection with T. gondii produces no readily observable
symptoms in healthy human adults. This asymptomatic state of infection is referred to as a
latent infection and has recently been associated with numerous subtle adverse
or pathological behavioral alterations in humans.In infants, HIV/AIDS patients, and others with weakened immunity,
infection can cause a serious and occasionally fatal illness, toxoplasmosis
T. gondii has been shown to alter
the behavior of infected rodents in ways thought to increase the
rodents' chances of being preyed upon by cats.
|
Vibrio anguillarum 鰻弧菌
|
Vibrio anguillarum is
a Gram-negative,
curved-rod bacterium with
one polar flagellum.
It is an important pathogen of cultured salmonid fish,
and causes the disease known as vibriosis or red pest of eels. The disease has been observed in salmon, bream, eel, mullet, catfish,
and tilapia,
amongst others. The organism is most prevalent in late summer in salt or brackish
water and transmission is
mainly horizontal by direct contact. It is widely distributed across the world.
Multiple haemorrhages in the body and skin changes
signifying systemic involvement occur. Splenomegaly (enlargement of spleen) may
be evident in young fish. Diagnosis relies on culture of V. anguillarum and the use
of monoclonal
antibodies.[1]
and control[edit]Various antibiotics such as ampicillin, chloramphenicol, nalidixic acid derivatives, nitrofurans, sulfonamides, and trimethoprim can be used to treat the fish. Resistance is emerging,
however. A vaccine against V. anguillarum is available. |
Vibrio harveyi 哈維弧菌
https://en.wikipedia.org/wiki/Vibrio_harveyi

|
Vibrio harveyi is a Gram-negative, bioluminescent, marine bacterium in the genus Vibrio. V. harveyi is rod-shaped,
motile (via polar flagella),
facultatively anaerobic, halophilic, and competent for both fermentative and
respiratory metabolism. It does not grow below 4 °C or above 35 °C. V. harveyi can be found
free-swimming in tropical marine waters, commensally in the gut microflora of marine animals, and as both a primary and opportunistic pathogen of marine animals, including Gorgonian corals, oysters, prawns, lobsters, the common
snook, barramundi, turbot, milkfish, and seahorses. |
|
It is responsible for luminous vibriosis, a disease that
affects commercially farmed penaeid prawns. Additionally, based on samples taken by ocean-going
ships, V. harveyi is
thought to be the cause of the milky seas effect,
in which, during the night, a uniform blue glow is emitted from the seawater. Some glows can cover nearly 6,000 sq mi (16,000 km2).sensing
Groups of V. harveyi bacteria communicate by quorum
sensing to coordinate the production of
bioluminescence and virulence factors. Quorum sensing was first studied in V. fischeri (now Aliivibrio fischeri), a marine bacterium that uses a synthase (LuxI) to produce a species-specific autoinducer (AI) that binds a cognate receptor (LuxR) that regulates
changes in expression. Coined "LuxI/R" quorum sensing, these systems have been
identified in many other species of Gram-negative bacteria.[3] Despite its relatedness to A.
fischeri, V. harveyi lacks a LuxI/R quorum-sensing system, and instead employs
a hybrid quorum-sensing circuit, detecting its AI through a membrane-bound histidine
kinase and using a phosphorelay to convert
information about the population size to changes in gene expression Since their identification in V. harveyi, such hybrid
systems have been identified in many other bacterial species. V. harveyi uses a second AI,
termed autoinducer-2 or
AI-2, which is unusual because it is made and detected by a variety of different
bacteria, both Gram-negative and Gram-positive. Thus, V. harveyi has been instrumental to the understanding and
appreciation of interspecies bacterial communication. |
CpG oligodeoxynucleotides
CpG寡脫氧核苷酸
https://en.wikipedia.org/wiki/CpG_Oligodeoxynucleotide
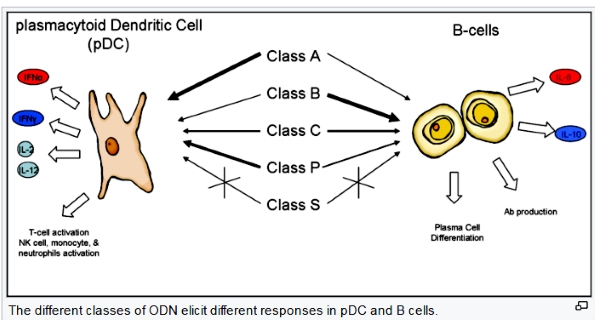
|
CpG
oligodeoxynucleotides (or
CpG ODN) are short single-stranded synthetic DNA molecules
that contain a cytosine triphosphate deoxynucleotide ("C")
followed by a guanine triphosphate deoxynucleotide ("G").
The "p" refers to the phosphodiester link
between consecutive nucleotides, although some ODN have a modified phosphorothioate (PS)
backbone instead. When these CpG motifs are
unmethylated, they act as immunostimulants.[1] CpG motifs are considered pathogen-associated molecular patterns (PAMPs) due to their abundance in microbial genomes but their rarity in vertebrate genomes. The CpG PAMP is recognized by the pattern recognition receptor (PRR) Toll-Like Receptor 9 (TLR9), which is constitutively expressed only in B cells and
plasmacytoid dendritic cells (pDCs)
in humans and other higher primates.
Since 1893, it has been recognized that Coley's
toxin, a mixture of bacterial cell lysate, has
immunostimulatory properties that could reduce the progression of some carcinomas but it was not until 1983 that Tokunaga et al.
specifically identified bacterial DNA as the underlying component of the lysate
that elicited the response Then, in 1995 Krieg et al. demonstrated that the CpG motif
within bacterial DNA was responsible for the immunostimulatory effects and
developed synthetic CpG ODN.[6] Since then, synthetic CpG ODN have been the focus of
intense research due to the Type I pro-inflammatory response they elicit and their successful use as vaccine adjuvants.Features[edit]Synthetic CpG ODN differ from microbial DNA in that they
have a partially or completely phosphorothioated (PS) backbone instead of the typical phosphodiester
backbone and a poly G tail at the 3' end, 5' end, or both. PS modification
protects the ODN from being degraded by nucleases such as DNase in the body and poly G tail enhances cellular uptake. The poly G tails form intermolecular tetrads that result
in high molecular weight aggregates. These aggregates are responsible for the
increased activity the poly G sequence impart; not the sequence itself.[8] Numerous sequences have been shown to stimulate TLR9 with
variations in the number and location of CpG dimers, as well as the precise base
sequences flanking the CpG dimers. This led to the creation of five unofficial
classes or categories of CpG ODN based on their sequence, secondary structures,
and effect on human peripheral blood mononuclear cells (PBMCs). The five classes are Class A (Type D), Class B (Type K),
Class C, Class P, and Class S. It is important to note that during the discovery process,
the "Classes" were not defined until much later when it became evident that ODN
with certain characteristics elicited specific responses. Because of this, most
ODN referred to in the literature use numbers (i.e., ODN 2006, ODN 2007, ODN
2216, ODN D35, ODN K3, etc.). The numbers are arbitrary and come from testing
large numbers of ODN with slight variations in attempts to find the optimal
sequence. In addition, some papers will give different names to previously
described ODN, complicating the naming convention even more.
|
DNA vaccination DNA疫苗接種
https://en.wikipedia.org/wiki/DNA_vaccination
DNA vaccination is
a technique for protecting against disease by injection with genetically engineered DNA so cells directly produce an antigen, producing a protective immunological response. DNA vaccines have potential advantages over
conventional vaccines, including the ability to induce a wider range of immune
response types.
Several DNA vaccines are available for veterinary use. One DNA vaccine has been approved for human
use. Research is investigating the approach for viral, bacterial and parasitic diseases in humans, as well as for several
cancers.
|
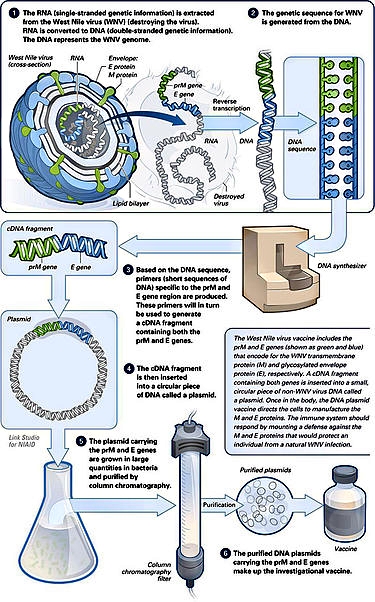
|
Mechanism of plasmids[edit]the plasmid inserts itself into the transfected cell nucleus, it codes for
a peptide string of a foreign antigen. On its surface cell the displays the
foreign antigen with both histocompatibility complex (MHC) classes I and class
II molecules. The antigen-presenting cell then travels to the lymph nodes and
presents the antigen peptide and costimulatory molecule signaled by T-cell,
initiating the immune response.[18]
insert design
Immunogens can be targeted to various cellular compartments to improve antibody
or cytotoxic T-cell responses. Secreted or plasma membrane-bound antigens are more effective at inducing
antibody responses than cytosolic antigens, while cytotoxic T-cell responses can be improved by targeting antigens
for cytoplasmic degradation and subsequent entry into the major histocompatibility complex (MHC) class I pathway. This is usually accomplished by the addition of N-terminal ubiquitin signals.[19][20][21]
The conformation of the protein can also affect antibody
responses. “Ordered” structures (such as viral particles) are more effective
than unordered structures. Strings of minigenes (or MHC class I epitopes) from different pathogens raise cytotoxic T-cell
responses to some
pathogens, especially if a TH epitope is also included. |
|
|
Diagnostic kit
檢驗試劑 |

|
Monoclonal antibody, Rapid test strip,
Test reagent kit for Avian , Swine , Bovine , Dog and
Feline diseases, etc. |
|
Links for references: (Welcome for recommending
helpful application links)
30 Awesome Puppy
Care Tips
List of epidemics ( https://en.wikipedia.org/wiki/List_of_epidemics )
History Of Pandemics

1. What Is A Pandemic, 2. List Of Pandemics Throughout The History, 3. The Deadliest Pandemics, 4. What Is a Pandemic Influenza, 5. Summary
|